GEMINI projects span a wide breadth of research areas and often involve multi-institutional collaborations. We encourage high-risk, high-reward projects with strong translational viability. We will fund unconventional approaches and new investigators to gastroesophageal cancer research who may have difficulty receiving funding from other sources.
GEMINI Investigators Have Expertise in Preclinical Studies
Individual patient-derived gastric and esophageal cancer organoids are sequenced and used in drug screening (Figure 1). Upon success in organoid models, genetically engineered mouse models of gastroesophageal cancer are used to test new therapies (Figure 2). Therapies showing success in these preclinical models can be taken to clinical trials.
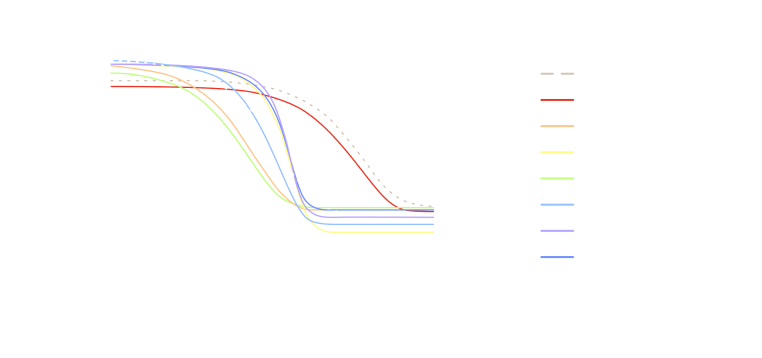
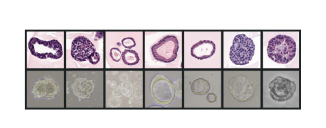
Figure 1: Individual patient-derived gastric cancer organoids can be generated from patient tumor biopsies, sequenced, and used in drug screening.
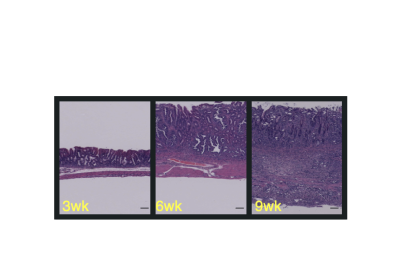
Figure 2: Genetically engineered mouse models of gastroesophageal cancer recapitulate the human disease and can be used in preclinical studies to test new therapies.
GEMINI Investigators Have Robust Clinical Trial Experience
GEMINI investigators have led clinical trials testing targeted therapies in combination with immunotherapy and chemotherapy. This new drug is an antibody targeting a protein that was newly identified as being overexpressed in gastroesophageal cancer (Figure 3).

Figure 3: Results from a Phase 2 Clinical trial using the antibody DKN-01 that targets the secreted protein Dickkopf-1 (DKK1) which is part of the Wnt/β-catenin pathway and is elevated in GE cancer. Patients with high DKK1 tumor expression treated with DKN-01, immunotherapy, and chemotherapy show improved clinical outcomes in GEJ or GC patients with advanced disease. SD, stable disease, PR, partial response, CR, complete response.
Examples of high-risk, high-reward funded projects include:
Dr. George Calin at MD Anderson Cancer Center is a pioneer in the field of small non-coding RNAs and cancer. Non-coding RNAs include micro RNAs (miRNA) and long non-coding RNAs (lncRNAs) that do not translate into proteins but have long been known to be important developmentally and are now gaining importance in cancer. Dr. Calin has identified microRNAs and long non-coding RNAs that are altered in human cancers and is investigating their role as potential therapeutic targets as well as signatures of diagnosis, prognosis, and response to treatment. Dr. Calin has developed novel approaches and compounds to target miRNAs in solid tumors. Dr. Calin's research has led to a multi-institutional collaboration with investigators at Columbia Medical Center and Case Western targeting a specific miR upregulated in gastric cancer.

Dr. Pradipta Ghosh at the University of California at San Diego leads a transdisciplinary team of scientists who create and validate disease maps that are geared to improve success rates in drug discovery. Built using high-throughput data analysis techniques, precision computational networks and Artificial Intelligence, these disease maps help identify universally conserved (hence, invariant) patterns of gene expression changes in diseases such as cancer. This approach can identify high-value therapeutic targets that can halt and/or reverse disease progression regardless of biological complexity and disease heterogeneity. It can also predict with precision a drug's potential benefit as well as the efficacy of combination therapies in diseases. Their work has identified 9 potential targets that are now being validated in pre-clinical patient-derived tumor organoids and mouse models of gastric cancer in multiple GEMINI-affiliated labs at UCSD, Columbia Medical Center, and Weil Cornell.
Investigators who join GEMINI will have access to all GEMINI affiliated projects and collaborative opportunities with associated researchers.